A common condition, endometriosis is a painful condition that can impact your everyday life. When you have endometriosis, tissue similar to the lining of the uterus grows in other places within your abdomen and pelvic area. Endometriosis can cause painful and heavy periods, as well as fertility issues.
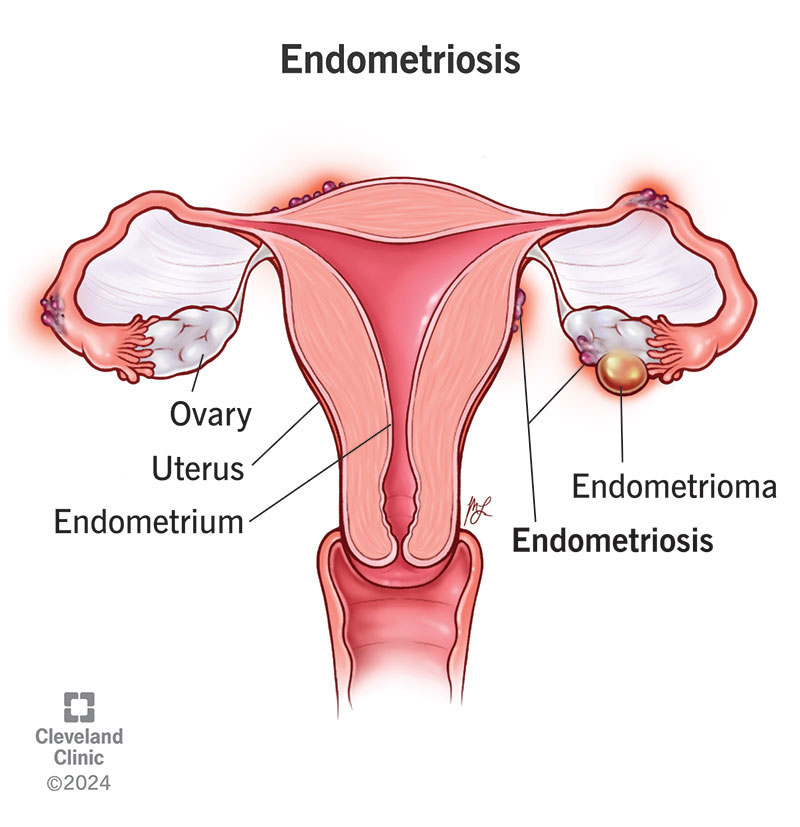
Endometriosis is a condition where tissue similar to the lining of your uterus grows on other parts of your body. When this tissue grows in the wrong places, it can cause you to experience uncomfortable symptoms that can impact your daily life. Some people with endometriosis also have issues getting pregnant.
The endometrium is the inner lining of your uterus. This tissue is what you shed during a menstrual period. Think of endometrium as layers of tissue that build up along the inside lining of your uterus. When you have a period, these layers fall away from the walls of your uterus and leave your body. If you get pregnant, the endometrium helps support the early phases of development.
When you have endometriosis, endometrial-like tissue grows on other organs or structures. This tissue can grow within your abdomen, pelvis or even chest. This tissue is hormonally sensitive and can become inflamed during your menstrual cycle. These areas of endometrial-like tissue can cause ovarian cysts, superficial lesions, deeper nodules, adhesions (tissue that connects your organs and binds them together) and scar tissue within your body.
A few places you can develop endometriosis include the:
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Endometriosis is a common condition that can interfere with your everyday life. It can cause long-term pain, disruptions to your menstrual cycle and fertility issues. The symptoms of endometriosis are often manageable with treatment.
Endometriosis is a condition that most commonly impacts people between the ages of 25 and 40. It can also happen to younger people during their teenage years. Although many people find relief from endometriosis symptoms after menopause, it can still cause discomfort and pain.
There are some factors that can place you at a higher risk of developing endometriosis. These factors can include:
Although experts don’t know the exact cause of endometriosis, they do see a connection between a family history of the condition and an increased risk of developing it at some point. If another person in your family — your mother, grandmother or sister — has endometriosis, talk to your healthcare provider about your risk.
The cause of endometriosis is unknown. When you have endometriosis, tissue similar to the lining of your uterus grows in the wrong places. When it develops in places like the outside of your uterus, fallopian tubes, ovaries, intestine and within your pelvic cavity, it can cause painful symptoms. This pain is related to increased inflammation and often fibrosis and adhesions.
When endometrial-like tissue grows outside of your uterus, it can cause scar tissue (adhesions). These sections of scar tissue can fuse your organs — creating connections between them that normally wouldn’t be there. This can lead to discomfort and pain.
There are many symptoms connected with endometriosis. The main symptom is pain. This pain can be intense or mild. It can typically be felt in your abdomen, pelvic region and lower back. Although endometriosis is a common condition, not all people will experience symptoms. Sometimes, you can have endometriosis and not know until it’s found during another procedure or investigation of infertility.
People who do experience symptoms of endometriosis may have:
There’s no connection between the symptoms of endometriosis and the severity of the condition. Some people may have very few patches of endometriosis and still experience severe pain. Other people might have severe endometriosis, but not experience a great deal of pain.
Many people experience pain during periods from endometriosis. This pain is often felt in your abdomen, lower back and pelvic area. Periods can also be heavier than typical, and there can be spotting (light bleeding) between cycles.
In many cases, an endometriosis diagnosis will start with your symptoms. Painful and heavy periods might cause you to reach out to your healthcare provider. Once at an appointment, your provider (typically an Ob-Gyn) may start by asking you for your personal medical history, about any previous pregnancies and if any other people in your family have endometriosis. Your provider may do a pelvic exam. If your healthcare provider needs more information they’ll likely perform pelvic imaging starting with an ultrasound. Depending on your symptoms, physical exam and ultrasound results, an MRI may also be ordered for further endometriosis mapping. A laparoscopy may be offered for both definitive diagnosis and treatment. It can be a useful way to confirm endometriosis because your surgeon doing the procedure can use a small camera (laparoscope) to look inside your body. A biopsy (small tissue sample) might be taken during this procedure. The biopsy will be sent to a lab to confirm the diagnosis.
Sometimes, you find endometriosis by accident. Not all people who have endometriosis will experience symptoms. In these cases, your provider might discover the condition during a different procedure.
Your healthcare provider will help create your treatment plan for endometriosis based on a few factors, including:
In many cases, your treatment plan will focus primarily on managing your pain and improving fertility issues (if you are planning on a future pregnancy). This can be done through medications and surgery.
Medications are often used to help control the symptoms of endometriosis. These can include pain medications and hormone therapies.
Hormonal options for suppressing endometriosis can include:
With all of these medications, it’s important to note that your symptoms can come back if you stop taking the medication. These medications aren’t recommended during pregnancy or if you are actively attempting to achieve pregnancy. Talk to your healthcare provider about the pros and cons of each medication before starting.
Medications for endometriosis pain relief can include:
In some cases, your provider might recommend surgery as a way to confirm and treat endometriosis. There are always risks to a surgical procedure. However, surgery for endometriosis can be an effective way to relieve pain and, in some cases, improve your fertility.
Endometriosis is considered a chronic disease. Many people experience relief from endometriosis pain after surgery, but the symptoms may return within a few years. The severity of your endometriosis could play a part in how quickly — if at all — it comes back after surgery. Your provider might suggest combining a surgical procedure with medications for the best outcome. Your provider may recommend pelvic floor physical therapy with or without medications for central nerve pain.
Surgical options to treat endometriosis include:
If you have endometriosis and are trying to achieve pregnancy, in vitro fertilization (IVF) may help you achieve this goal.
In some cases, endometriosis can go away on its own. Over time, endometriosis lesions can occasionally get smaller, and you may have fewer of them. This can also happen after menopause, which is often related to a drop in the amount of estrogen in your body.
For many people, endometriosis needs to be continuously treated to control symptoms like pain. It’s important to maintain a regular appointment schedule with your healthcare provider so that you can work together on managing your condition long term.
Over time, the endometrial-like tissue that grows outside of your uterus can cause cysts, adhesions and scar tissue. This can cause you to experience long-term (chronic) pain — especially during menstrual periods. Many people with endometriosis may also have difficulties getting pregnant. Treatment can sometimes help with this issue.
As you age and go through menopause, the symptoms of menopause may improve. This is related to the hormonal changes your body goes through during menopause.
Endometriosis isn’t a condition you can necessarily prevent. There are certain factors that can reduce your risk of developing the condition, but in some cases, you may still have endometriosis. There could be a genetic reason that some people develop endometriosis. If other people in your family (mother or grandmother) have been diagnosed with endometriosis, talk to your provider about your risk of also developing the condition.
A few factors that can reduce your risk of endometriosis include:
There are several medical complications that can happen if you have endometriosis. People with endometriosis may experience fertility issues (difficulty getting pregnant). This can sometimes be helped with treatment options like medications, surgery for endometriosis or fertility treatments like IVF.
People with endometriosis can also experience bowel or bladder issues. These may include pain with voiding or with bowel movements, or seeing blood in your urine (pee) or stool (poop). With severe endometriosis of your ureter (the tube that carries urine from your kidney to your bladder), you can occasionally get swelling of your kidney. Endometriosis can occasionally impact your lung or diaphragm, which can lead to shortness of breath, chest pain or lung collapse during menstrual cycles. Chronic (long-term) pain is another issue related to endometriosis. Your healthcare provider will work with you to manage these issues to improve your daily life.
Endometriosis is associated with a small increased risk of developing epithelial ovarian cancer, mainly including clear cell and endometroid carcinomas. This risk is extremely low and no preventative screening is currently recommended.
You can get pregnant if you have endometriosis. However, people with endometriosis can have a difficult time getting pregnant. This condition can be a common cause of infertility. If you have endometriosis and want to get pregnant, talk to your healthcare provider about the best treatment option for you. You might need to change your medication or, in some cases, pursue a surgical option to treat your endometriosis. Your provider will work with you to find the best treatment plan to help support a pregnancy.
Menopause is a time of major change in your body. One thing that happens during this transition is a change in the levels of hormones in your body, specifically estrogen. There’s a link between your reproductive hormones and endometriosis. After menopause, with decreased estrogen levels, endometriosis lesions often decrease. This can also mean that you no longer experience symptoms of the condition or that they’re less intense than before menopause.
However, if you take hormones as a treatment for any symptoms you experience during menopause, your endometriosis may still cause symptoms.
A note from Cleveland Clinic
Endometriosis can cause you to experience long-term (chronic) pain, heavy periods and difficulties getting pregnant. You can manage these symptoms by working with your healthcare provider. If you notice any symptoms of endometriosis or are having unusual or painful periods, talk to your provider. There are treatment options that can help improve your daily life and manage your endometriosis over the long haul.
Last reviewed by a Cleveland Clinic medical professional on 07/27/2022.
Learn more about our editorial process.
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy