While the news is promising, check with your healthcare provider first
A: On August 12, 2021, the FDA updated its emergency use authorization for both Pfizer-BioNTech’s and Moderna’s COVID-19 vaccines for immunocompromised individuals, specifically, solid organ transplant recipients (kidney, liver, intestines, heart, lung and pancreas) or those who are diagnosed with conditions considered to be at the same level of immunocompromise.
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Transplant recipients have to be very careful because their immune systems are suppressed to reduce the chances of their bodies rejecting the new organs they receive. Overall, immunocompromised people are at increased risk for poor outcomes from COVID-19, and studies have shown a reduced antibody response for this patient population after vaccination.
On October 20, 2021, the FDA amended the EUA to include booster shots of all three vaccines (Pfizer, Moderna and Johnson & Johnson). For Pfizer and Moderna, the parameters are patients:
Additionally, the Johnson & Johnson vaccine booster was cleared for all recipients age 18 and up who received the single dose that one-shot vaccine. The FDA is also allowing a “mix and match” system for boosters. In other words, once you’ve completed your original vaccine dose, you can receive any of the available vaccines as a booster.
In July, the Centers for Disease Control and Prevention’s (CDC) Advisory Committee on Immunization Practices (ACIP) presented “Data and Clinical Considerations For Additional Doses In Immunocompromised People” during a meeting. One finding in this presentation was that recent studies have indicated that additional COVID-19 vaccine doses in immunocompromised people enhanced antibody responses and increased the proportion that responded. In smaller studies, the immunological responses of the third dose were about the same as prior doses.
Currently, the CDC recommends that moderately to severely immunocompromised people receive an additional dose. This includes people who have:
If you qualify, you can get the third dose of a COVID vaccine as soon as possible. However, talk to your healthcare provider first to determine if you should.
— Hematologist Matt Kalaycio, MD
Learn more about our editorial process.

The short answer: It’s complicated, but the basic care precautions still prevail, like washing your hands and isolating if you’re sick

Any large social gathering — from a family birthday party to an indoor music concert — has the potential to spread serious infection

It’s important to connect with a healthcare provider, get quality sleep and balance your activities with your energy levels

Just like the flu, COVID-19 will continue to evolve every year

It’s best to treat flu-like symptoms as if you have COVID-19

The duration varies, but symptoms can linger for a few days up to a couple weeks or more

Plan early — getting the right vaccines can help you stay healthy on your travels
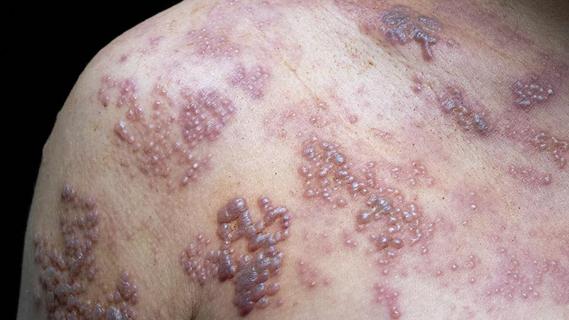
It’s 97% effective in preventing shingles in people between the ages of 50 and 69

Your metabolism may torch 1,300 to 2,000 calories daily with no activity

A gentle touch in all the right places may help drain your sinuses