Congestive heart failure is a long-term condition that happens when your heart can’t pump blood well enough to give your body a normal supply. Blood and fluids collect in your lungs and legs over time. Medications and other treatments help manage symptoms like swelling. Congestive heart failure is life-limiting for many.
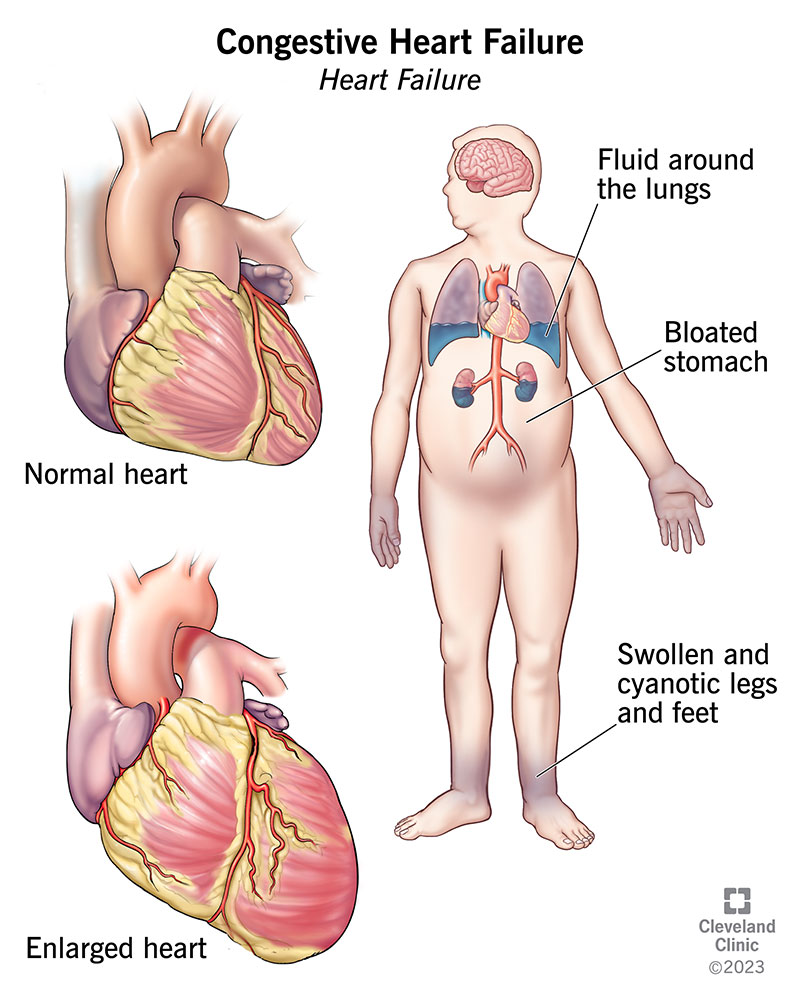
Congestive heart failure, or heart failure, is a long-term condition in which your heart can’t pump blood well enough to meet your body’s needs. Your heart is still working. But because it can’t handle the amount of blood it should, blood builds up in other parts of your body. Most of the time, it collects in your lungs, legs and feet.
Think of it like a shipping department that can’t keep up with getting all the shipments where they need to go. The shipping department is always running behind and things pile up. When things pile up, they cause issues.
Types of congestive heart failure include:
More than 6 million people in the United States have congestive heart failure. It’s the leading cause of hospitalization in people older than 65.
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
Congestive heart failure symptoms include:
Sometimes, you may have mild symptoms of congestive heart failure or none at all. This doesn’t mean you don’t have heart failure anymore. Symptoms of heart failure can range from mild to severe and may come and go.
Unfortunately, congestive heart failure usually gets worse over time. As it worsens, you may have more or different signs or symptoms.
Causes of congestive heart failure include:
Left-sided heart failure is the most common cause of right-sided heart failure. When your left ventricle isn’t working correctly, it allows blood to back up. At some point, this backup affects your right ventricle. Other causes include certain lung problems and issues in other organs.
Risk factors for congestive heart failure include:
Some of the complications from congestive heart failure include:
Your healthcare provider will ask you about your symptoms and medical history. They may ask you about:
You’ll also have a physical exam. Your provider will look for signs of congestive heart failure and diseases that may have made your heart muscle weak or stiff.
Heart failure is a chronic condition that gets worse with time. There are four heart failure stages (Stages A, B, C and D). They range from having a high risk of developing heart failure to having advanced heart failure.
Stage A (pre-heart failure) means you’re at a high risk of developing heart failure because you have a family history of congestive heart failure or you have one or more of these medical conditions:
Stage B (pre-heart failure) means your left ventricle isn’t working well and/or is structurally abnormal but you’ve never had symptoms of heart failure.
People with Stage C heart failure have a congestive heart failure diagnosis and currently have or previously had signs and symptoms of the condition.
People who have Stage D HFrEF (heart failure with reduced ejection fraction) have advanced symptoms that don’t get better with treatment. This is the final stage of heart failure.
Common tests to identify congestive heart failure, its stage and its cause include:
Your treatment will depend on the type of heart failure you have and, in part, what caused it. Medications and lifestyle changes are part of every heart failure treatment plan. Your healthcare provider will talk to you about the best treatment plan for you.
There’s no cure for heart failure. As congestive heart failure gets worse, your heart muscle pumps less blood to your organs, and you move toward the next stage of heart failure. Since you can’t move backward through the heart failure stages, the goal of treatment is to keep you from moving forward through the stages or to slow down the progression of your heart failure.
Treatment for people with Stage A heart failure includes:
Treatment for people with Stage B heart failure includes:
Treatment for people with Stage C HFrEF includes:
If the treatment improves or stops your symptoms, you still need to continue treatment to slow the progression to Stage D.
Treatment for people who have Stage D heart failure includes treatments for Stages A, B and C. In addition, it includes evaluation for more advanced treatment options, including:
Treatment for people with Stage C and Stage D heart failure and preserved EF (HFpEF) includes:
It’s very important for you to manage your other health conditions, such as:
Some conditions have signs and symptoms similar to congestive heart failure. If you have new or worsening nonurgent symptoms, tell your healthcare provider.
Complications of congestive heart failure treatments may include:
Although you can’t change some risk factors like age, family history or race, you can change your lifestyle to give yourself the best chance of preventing heart failure. Things you can do include:
With the right care, congestive heart failure won’t stop you from doing the things you enjoy. Your prognosis, or outlook for the future, will depend on:
One study says that people with congestive heart failure have a life span 10 years shorter than those who don’t have heart failure. Another study showed that the survival rates of people with chronic heart failure were:
A different study found that people who had heart failure had expected life spans ranging from three to 20 years after their hospital stay, depending on various factors like age and assigned sex at birth. It’s important to look at your specific situation when considering your prognosis.
Congestive heart failure is a chronic, or lifelong, condition. You’ll need to treat it for the rest of your life.
With the right care and treatment plan, many adults still enjoy life even though congestive heart failure limits their activities.
You can care for yourself by:
Congestive heart failure is a chronic, long-term illness that can get worse quickly, so it’s a good idea to let your provider and family know your preferences for medical care. You can complete an advance directive or living will to let everyone involved in your care know what you want. A living will details the treatments you want or don’t want to prolong your life. It’s a good idea to prepare a living will while you’re well in case you aren’t able to make these decisions at a later time.
People with congestive heart failure may need to limit how much salt and/or fluid they consume each day. Your provider can give you guidelines for this.
It’s important to let your provider know if you have new symptoms or if your heart failure symptoms get worse. Contact them for new or worsening:
You need emergency treatment when your heart failure suddenly gets worse. Providers call this acute decompensated heart failure. They can give you medicine and oxygen to improve your condition.
Questions to ask your healthcare provider include:
A note from Cleveland Clinic
If you have heart failure, you can take steps to improve your heart health. Take your medications, follow a low-sodium diet, stay active, take notice of sudden changes in your weight, keep your follow-up appointments and track your symptoms. If you have questions or concerns about your medications, lifestyle changes or any other part of your treatment plan, talk to your provider. They’re there to help you manage your heart failure.
Last reviewed by a Cleveland Clinic medical professional on 03/10/2023.
Learn more about our editorial process.
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy