Locations:

Potential benefits include fewer pesticides and insecticides and more nutrients — but it all comes at an additional cost

With anti-fatigue, anti-aging and anti-cancer claims, this fungi still needs more studies

Legality, toxicity and effectiveness are all ongoing concerns

This herbal supplement comes with health risks, especially if you take antidepressants
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy

Ordinary soap and water or an alcohol-based sanitizer can do the job
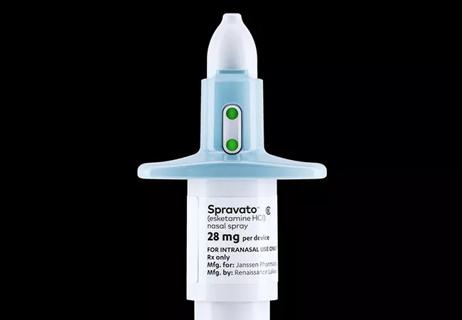
The possible benefits and risks, explained
Advertisement
Advertisement