The latest vaccine offers the most up-to-date protection
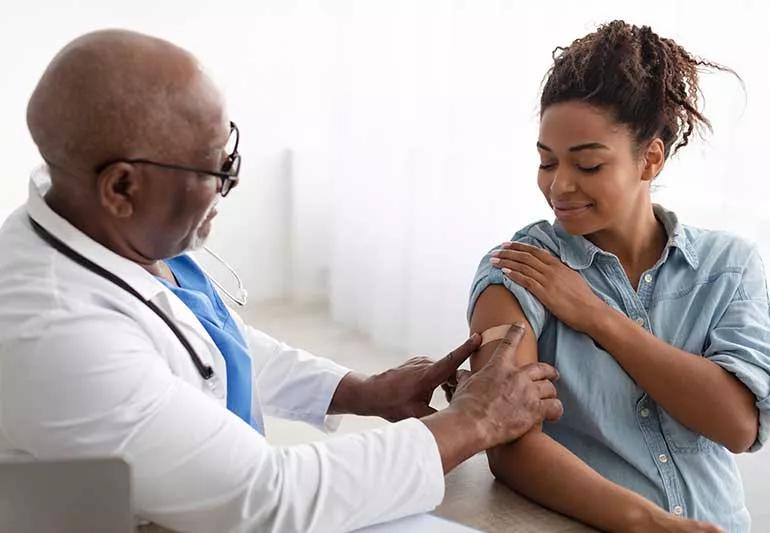
Viruses change all the time in an effort to do one simple thing: Bypass your body’s defensive immune system and make copies of itself so it can infect others and survive.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
We see this happen with influenza (the flu). And every year, we design flu vaccines that can fight off new and dominant strains of the virus to minimize its spread and the severity of symptoms associated with the flu.
Coronaviruses are no different: They’re the latest in a long line of respiratory viruses that continue to evolve and peak during the fall and winter seasons.
To prepare for this year’s respiratory season, the U.S. Centers for Disease Control and Prevention (CDC) has announced the release of a new, updated COVID-19 vaccine. This vaccine specifically targets current circulating variants of the virus, including XBB.1.5, EG.5.1 and FL.1.5.1.
So, what does that mean if you already got the previous bivalent omicron booster? And what if you or your child has never had a COVID-19 vaccine before? Infectious disease specialist Ryan Miller, DO, and occupational health specialist Craig Thorne, MD, explain how the updated vaccine works and who should get it as soon as possible.
The new 2023 COVID-19 vaccine is different from bivalent boosters of the past because it stands on its own as a way of protecting against current variants of the virus. Similar to vaccines that protected against the original virus, this updated vaccine is an mRNA monovalent vaccine that contains a single component that corresponds to a specific variant of the virus.
Advertisement
In simpler terms, according to the CDC, this means the new vaccine is designed to be the only effective way to protect against current circulating variants.
“The new 2023 vaccine includes more recent COVID-19 strains in its mRNA,” explains Dr. Miller. “They’re more up-to-date and should provide current immunity because the effectiveness of the bivalent vaccine was waning.”
In fact, the U.S. Food and Drug Administration (FDA) has shifted away from calling this a booster, instead referencing it as an updated COVID-19 vaccine. “This change in wording reflects that we have begun treating COVID-19 like we treat the flu, with an annual vaccination,” says Dr. Thorne. “We encourage people to get their ‘annual flu shot,’ not a ‘flu booster.’ Calling it an updated COVID-19 vaccine also reflects that we are not just boosting existing immunity from previous vaccinations — this vaccine builds a new immune response to COVID-19 variants that are currently circulating.”
If you or your children belong in any of the following categories, you should get at least one dose of the updated 2023 vaccine as soon as possible:
“You’re not up-to-date on COVID-19 vaccination unless you receive this new COVID-19 vaccine,” states Dr. Miller. “Even if you’ve had a COVID vaccine in the last six months, you’re not considered up-to-date until you have this new vaccine.”
According to the CDC, the following guidelines are helpful to determine whether or not you’re up-to-date on current COVID-19 vaccination:
If you’re older than 65 or are immunocompromised, you may also get additional updated vaccines as-needed:
Advertisement
COVID-19 is still dangerous. If you’re ever concerned about or unsure of your COVID-19 immunity, making an appointment with a healthcare provider can provide some much-needed clarity before you find yourself in peak respiratory season.
And now that we’re able to get an updated COVID-19 vaccine alongside frequently updated flu vaccines, as well as a new vaccine for RSV (respiratory syncytial virus), we can begin to better protect ourselves and others during critical times for infections.
“The approach to the most current version of the COVID-19 vaccine is similar to how we approach updated flu vaccines every year,” clarifies Dr. Miller. “We anticipate that COVID-19 vaccine rollouts will turn into what our current flu vaccination campaigns are now — every fall, we’ll offer a new, updated version of the vaccine.”
Advertisement
Learn more about our editorial process.
Advertisement

Children as young as 6 months should get vaccinated, but dosage guidelines depend on kids’ ages and past vaccines

Updated vaccinations are recommended to better protect against the evolving virus

Redness, swelling, itching and rash can happen when your body’s immune system reacts to the vaccine injection
Irregularities in cycle length and flow aren’t a cause for concern

Before you panic, here are the options to consider

Our expert explains why swollen lymph nodes happen

Covering your mouth when you cough and staying home when you’re sick are a couple ways to help keep yourself and others COVID-free

The virus often clears up on its own within two years, but it can also linger and can cause further complications

If you’re feeling short of breath, sleep can be tough — propping yourself up or sleeping on your side may help

If you fear the unknown or find yourself needing reassurance often, you may identify with this attachment style

If you’re looking to boost your gut health, it’s better to get fiber from whole foods