It’s an FDA-approved treatment for urinary incontinence and overactive bladder
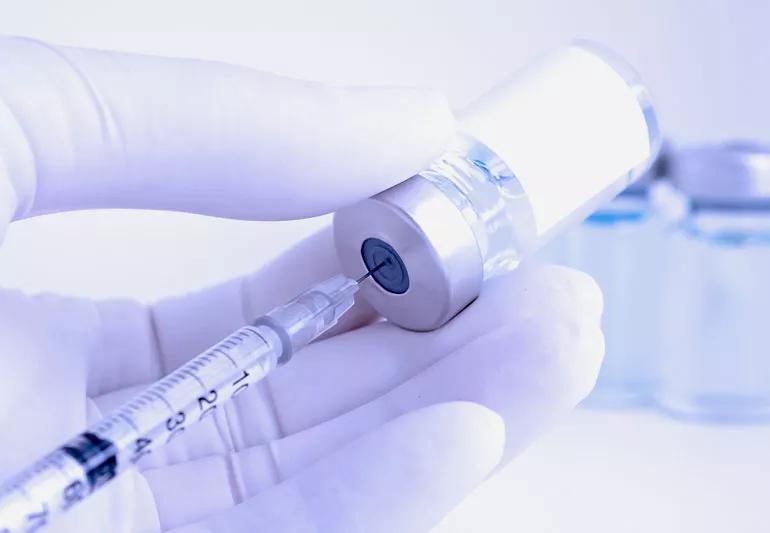
It’s happened to most of us at least once: You cough, laugh or run … and a little pee comes out. While a bit embarrassing, rare leaking episodes aren’t a big deal.
Advertisement
Cleveland Clinic is a non-profit academic medical center. Advertising on our site helps support our mission. We do not endorse non-Cleveland Clinic products or services. Policy
But what if it’s happening a lot? What if the leaking is impacting how you live your life?
Well. That’s a problem.
The same goes for urgency. Waking up at night to pee — or getting a sudden, barely controllable urge to go — isn’t concerning if it’s a rare occurrence.
But if you’re waking up every night, several times a night because you need to pee? If you’re having to stop on the side of the road because you can’t wait 10 more minutes until you get home? If you feel like your life revolves around your unpredictable bladder?
Yeah. That’s an issue.
Incontinence and overactive bladder (OAB) are common conditions. And luckily, they have multiple causes and available treatment options. If more traditional methods aren’t getting the job done, your doctor may suggest a surprising treatment: Botox® (onabotulinumtoxinA) injections.
We spoke to urologist Sandip Vasavada, MD, to learn more about the procedure, how it works and how safe it is.
Most people think Botox is a tool for plastic surgeons and not urologists. But the drug is approved by the U.S. Food and Drug Administration (FDA) for people (including children) with overactive bladder symptoms with no apparent neurologic cause.
Advertisement
Research shows the drug is 70% to 80% effective for people who have sudden urgency-related leakage or incontinence, says Dr. Vasavada.
While effective, Botox isn’t a go-to treatment. Your doctor will probably recommend pelvic floor therapy, bladder retraining, medications, and behavioral and lifestyle changes before suggesting an invasive procedure.
While it’s primarily used for bladder issues without a neurologic cause, Botox is occasionally used to address incontinence connected to neurological conditions like multiple sclerosis (MS) or a spinal cord injury.
While it’s a safe procedure, bladder injections of this sort aren’t for everyone. Your doctor may recommend against Botox if:
Botox chemically affects muscle-nerve connections. That’s why it can also be used to treat other disorders, including chronic migraine and severe underarm sweating.
“The drug decreases the spasms that cause overactive bladder symptoms,” Dr. Vasavada explains. “It’s not so much for frequency of urination, as much as that sudden urge to go. It’s good to help people when urination is something they can’t postpone without leakage occurring.”
Botox injections into the bladder are usually outpatient or office-based procedures. It doesn’t require general anesthesia, and you don’t need anybody to drive you to or from your appointment.
Your urologist first injects a local anesthetic — similar to the numbing medication you get when having a cavity filled. After giving the anesthetic time to take effect, your doctor will spend 15 minutes making a series of small injections into the wall of your bladder through a lighted tube called a cystoscope.
After completing the injections, your doctor will keep you under observation for 30 minutes to make sure you’re alright. Before you leave, you’ll be asked to use the restroom, so your doctor can ensure that you’re able to completely empty your bladder unassisted. You may also get a prescription for a short-term antibiotic. Taking that medication should protect you against infection.
Advertisement
All told, you’ll spend approximately one hour at the doctor’s office.
It’s important to note that the injections won’t take immediate effect. While some people notice a difference within a few days, people with urinary incontinence can expect to wait one to two weeks before seeing the number of daily leakage episodes come down. According to the makers of Botox, you should expect to see “full results” by week 12.
It’s also important to remember that this treatment isn’t a cure. You may still experience occasional issues with urinary incontinence or overactive bladder, and the effects of the injections will wear off over time and need to be repeated. More on that later.
The main side effects of the procedure are urinary retention, urinary tract infection, and painful or difficult urination.
The most common side effect, according to the makers of Botox, is urinary tract infection (UTI). They report that 18% of patients experience this side effect.
The makers of Botox report that approximately 9% of people who receive bladder injections experience painful or difficult urination.
Urinary retention is a temporary inability to fully empty your bladder on your own. This side effect occurs in about 5% to 8% of patients.
Advertisement
Those people may need temporary catheterization to guard against an increased risk of urinary tract infections, Dr. Vasavada notes. And for that reason, some people decide against the procedure.
“We just can’t predict who’s likely to have emptying problems,” he says. “It’s not a high likelihood, but not zero either. We can’t predict who will experience this side effect.”
But most people haven’t had that hesitancy, according to Dr. Vasavada. “Since it’s been approved for incontinence, it’s certainly generating more enthusiasm and interest.”
Botox has become so common that we sometimes forget what it’s made of.
Clostridium botulinum is the bacteria that causes botulism, a rare and dangerous infection that can cause paralysis and death.
While it’s extremely rare, it’s possible for Botox to migrate to other parts of your body. If you have any of the botulism symptoms below, you should call 911 immediately:
Dr. Vasavada says that Botox is frequently effective, as long as people repeat the treatment roughly once every six months.
Advertisement
“It does wear off. Otherwise, it’s pretty well-tolerated and works a good majority of the time,” he adds.
But in the event that you’re seeing symptoms return earlier than expected, you should contact your doctor. You may need to receive more frequent treatments.
Learn more about our editorial process.
Advertisement
Most recommended precautions center around minimizing bruising or swelling

With repeat injections over time, you may be able to slow the development of new wrinkles
The cosmetic injection may help train your muscles out of frowning, but there’s no hard data to say for sure
One reduces wrinkles by relaxing muscles; the other adds volume and smooths skin
They both treat fine lines and wrinkles, but how they’re made and how long results last differ
The big difference with this procedure is the smaller dosing
Both are used to treat fine lines and wrinkles
If you're tired of products that don’t stop your serious sweating, Botox could be life-changing

Wearing a scarf, adjusting your outdoor activities and following your asthma treatment plan can help limit breathing problems

Your diet in the weeks, days and hours ahead of your race can power you to the finish line

When someone guilt trips you, they’re using emotionally manipulative behavior to try to get you to act a certain way